Bond length of H H is 0.64 and the bind length of F2 is 1.2. Electronegativities of H and F respectively are 2.1 and 4.1.What is the bond length of HF? 1)0.64 2)0.92 3)0.82 4)0.62
By A Mystery Man Writer
Bond length of H H is 0.64 and the bind length of F2 is 1.2. Electronegativities of H and F respectively are 2.1 and 4.1.What is the bond length of HF? 1)0.64 2)0.92 3)0.82 4)0.62
Bond length of H-H is 0-64 and the bind length of F2 is 1-2- Electronegativities of H and F respectively are 2-1 and 4-1-What is the bond length of HF- 1-0-64 2-0-92 3-0-82 4-0-62

Oxygen reduction electrochemistry at F doped carbons: A review on the effect of highly polarized C-F bonding in catalysis and stability of fuel cell catalysts - ScienceDirect

Patai S., Rappoport Z. (Eds.) - The Chemistry of Organic Selenium and Tellurium Compounds. v.1, PDF, Functional Group

Rare-earth-containing perovskite nanomaterials: design, synthesis, properties and applications - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C9CS00330D

Actinides - Chemistry and Phys. Properties - Structure & Bonding v.59 (1985) Pp.1-298, PDF, Electron Configuration

Which of the following has the minimum bond length ?

Chapter 1-5 PDF, PDF, Neutron
9.10: Bond Energies and Bond Lengths - Chemistry LibreTexts
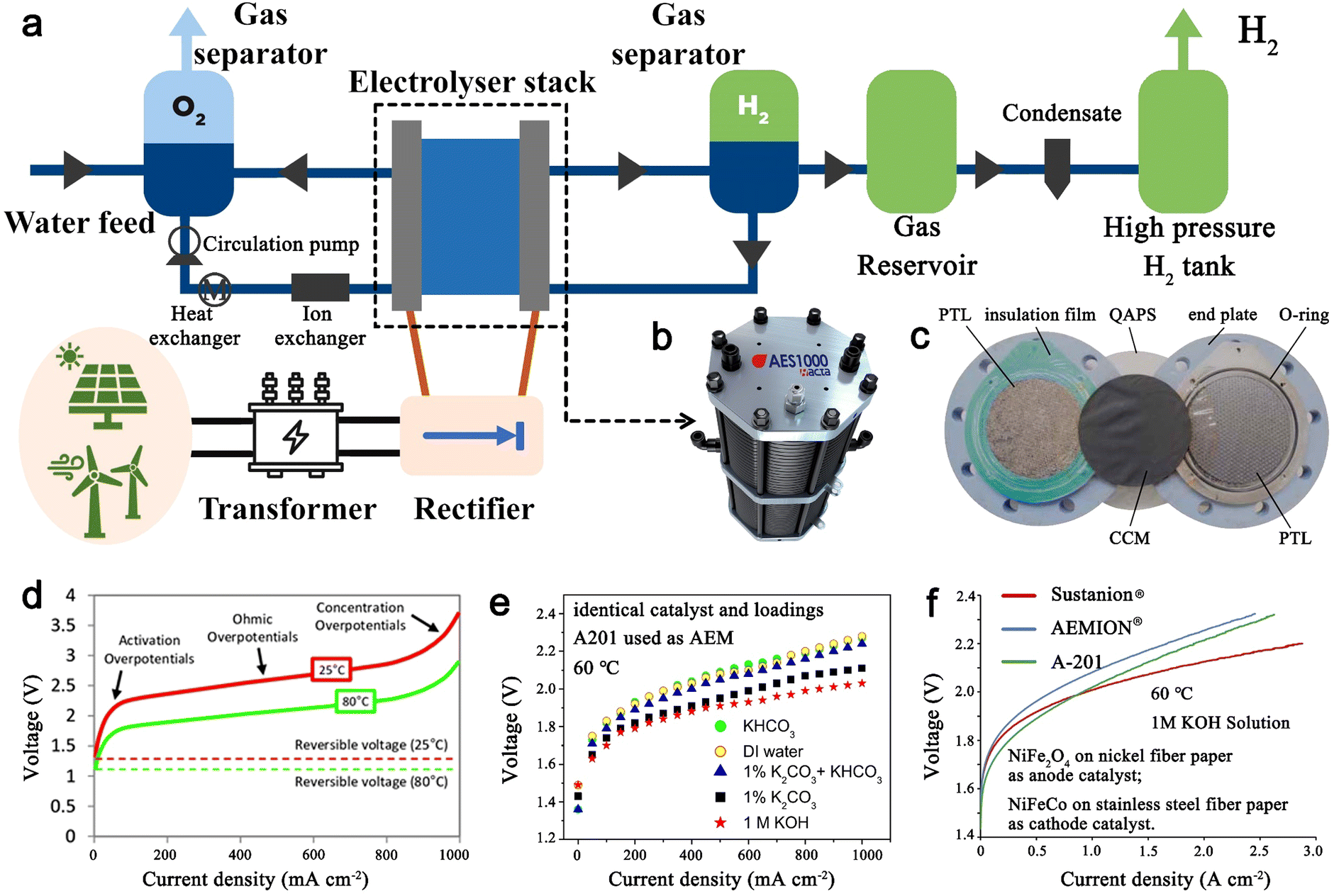
Anion-exchange membrane water electrolyzers and fuel cells - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D2CS00038E

Chemical Engineering Reviewer Edited, PDF, Molecules
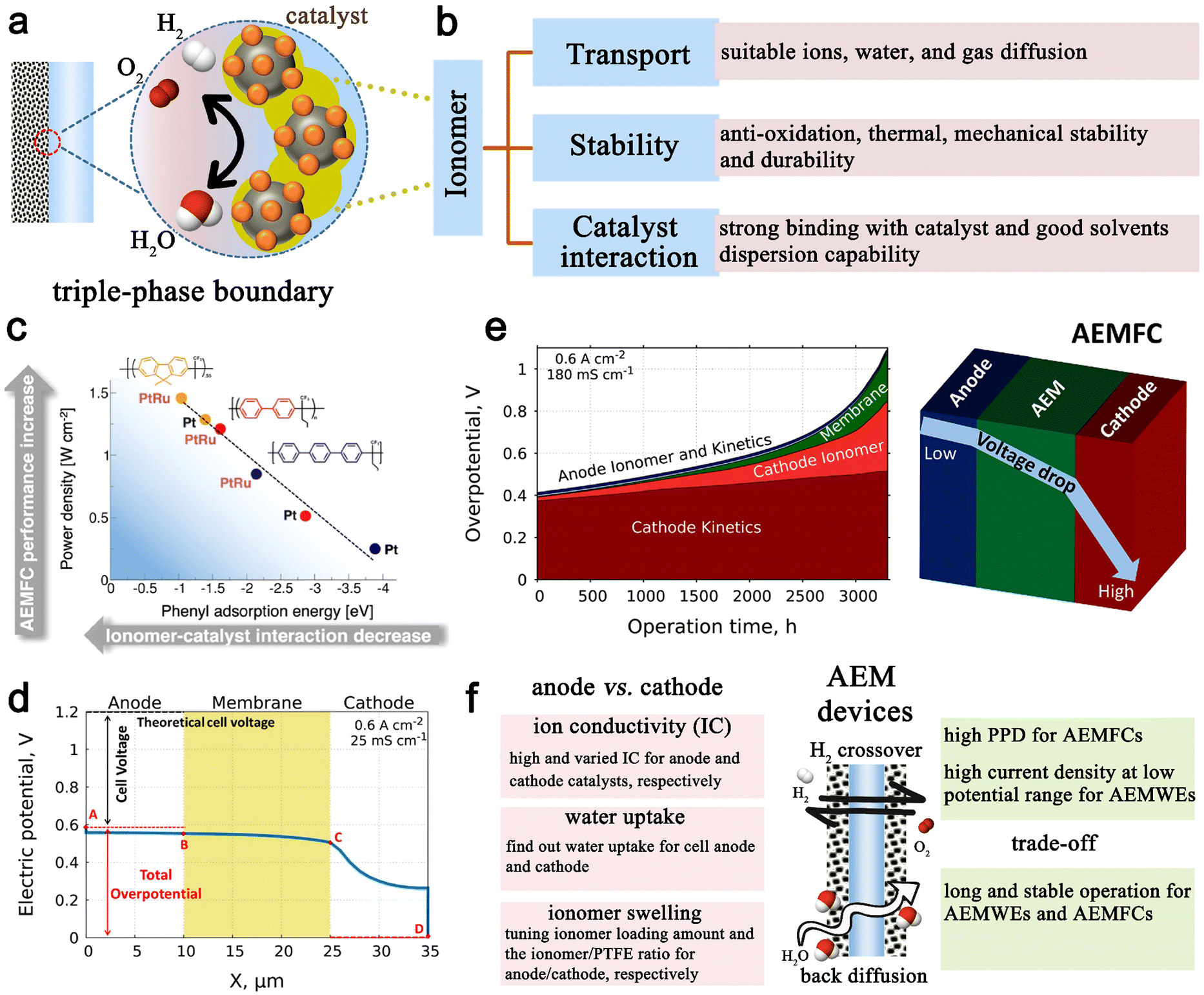
Anion-exchange membrane water electrolyzers and fuel cells - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D2CS00038E