Palette Life Sciences Announces FDA 510(k) Clearance for Barrigel® Rectal Spacer, Proven Safe and Effective at Minimizing the Harmful Long-Term Side Effects of Prostate Radiation Therapy - Palette Life Sciences

By A Mystery Man Writer
Groundbreaking technology introduces increased control to achieve optimal coverage proven to significantly reduce the risk of toxicity to the rectum SANTA BARBARA, CALIF. / STOCKHOLM, SWEDEN – June 9, 2022— Palette Life Sciences, a fully-integrated global life sciences company dedicated to improving patient outcomes, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance of […]

Palette Life Sciences Announces Publication of Barrigel® Pivotal

CAROL THRONDSON on LinkedIn: Palette Life Sciences Announces FDA 510(k) Clearance for Barrigel® Rectal…

Helena Jansson på LinkedIn: Palette Life Sciences Announces FDA

Palette Life Sciences Announces FDA 510(k) Clearance for Barrigel
Prostate Cancer Radiation Side Effects
SpaceOAR - Augmenix, Boston Scientific, and Conflicts of Interest

Optimization of polyethylene glycol-based hydrogel rectal spacer
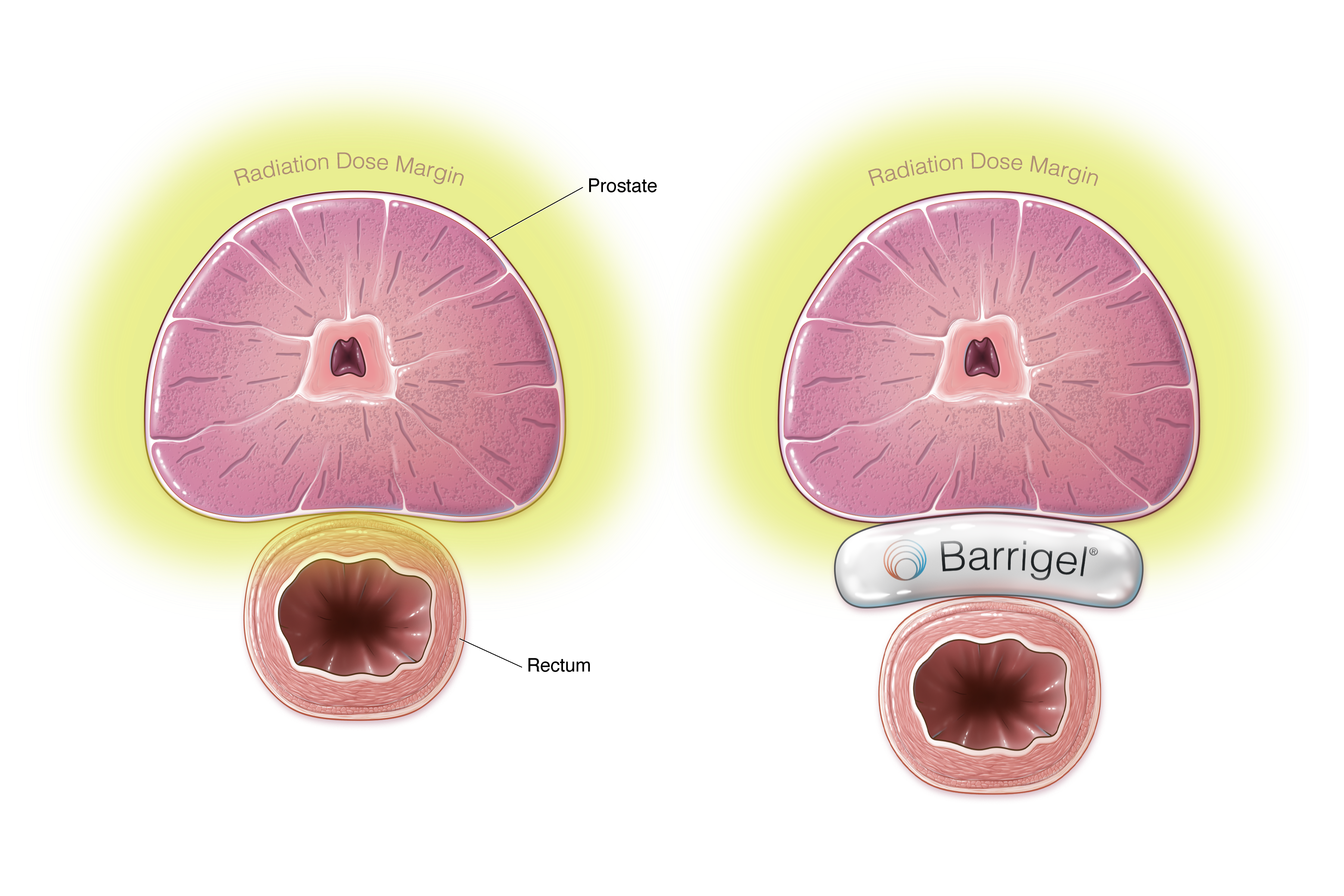
Barrigel® Rectal Spacing
Barrigel Proven Safe & Effective at Minimizing Side Effects

FDA clears BioProtect Balloon for rectal protection during

Rick Wymer on LinkedIn: Palette Life Sciences Announces FDA 510(k

FDA OKs Palette Life Sciences' Barrigel Rectal Spacer
Prostate Cancer Radiation Side Effects
SpaceOAR - Augmenix, Boston Scientific, and Conflicts of Interest
- 100 PACK Inline Axle ALUMINUM SPEED MICRO SPACERS for 8mm Axles RED Spacer

- 12mm spacer for LED size 3mm(T-1) & 5mm(T-1 3/4) [6452] : Sunrom Electronics

- Applying Interphase Spacers to Control Conductor Galloping

- James Leech on LinkedIn: Palette Life Sciences Announces FDA 510(k) Clearance for Barrigel® Rectal…
- 10PCS Inline Skate Wheel Bearing Spacer Aluminium Alloy Spacers

- Ready Stock / 100% Genuine] Set Calvin Klein Modern Cotton Lightly Lined With Foam (Gray)
- Black V-Strap Thong Sandals - CHARLES & KEITH UK

- Comfortable Leather Studded Flip Flops For Men And Women Perfect For Summer Beach Days From New_shoesstore, $28.14

- Com preços a partir de R$ 25, Zara abre loja com novo conceito no Brasil

- Premium Photo Vintage Retro style feshion clothing 1970s fashion Bellbottoms 70s hipsters bell bottom pants frayed jeans midi skirts maxi dresses tiedye peasant blouses and ponchos


