An ideal gas initially P_i ,V_i , and T_i is taken through a cycle

By A Mystery Man Writer
Click here:point_up_2:to get an answer to your question :writing_hand:an ideal gas initially at pi vi and ti is taken through a cycle
Click here👆to get an answer to your question ✍️ An ideal gas initially P-i -V-i - and T-i is taken through a cycle as shown in Figure- -a- Find the net work done on the gas per cycle 1-00 mol of gas initially 0-0C- -b- What is the net energy added by heat to the gas per cycle

Solved) - A 1.00-mol sample of a monatomic ideal gas is taken through the - (1 Answer)

SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through a cycle as shown below. (Let the factor n 3.6.) nP; P; nV; (a Find the net work done

Relating Pressure, Volume, Amount, and Temperature: The Ideal Gas Law

Thermodynamics problems

PPT - More Thermodynamics PowerPoint Presentation, free download - ID:4461196


11/14/2013PHY 113 C Fall Lecture 221 PHY 113 C General Physics I 11 AM – 12:15 PM MWF Olin 101 Plan for Lecture 22: Chapter 21: Ideal gas equations. - ppt download
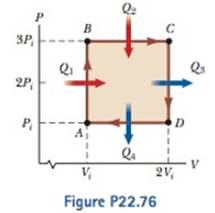
A 1.00-mol sample of a monatomic ideal gas is taken through the cycle shown in Figure P22.76. At point A, the pressure, volume, and temperature are P i , V i

Thermodynamics: An Engineering Approach - 5th Edition - Part II by 黑傑克 - Issuu
- PI ideal

- Buy Pi Mathematical Print Mathematics Wall Art Maths Student Gift Idea Pi Dorm Decor Single Print 970 INSTANT DOWNLOAD Online in India

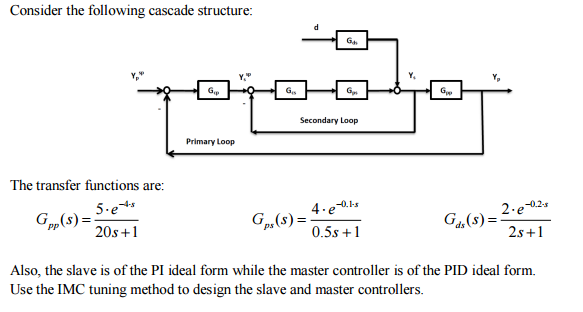
- Consider the following cascade structure: The
- The Numbers of Pi – Bad Idea T Shirts

- Pi Mathematical Print Mathematics Wall Art Maths Student Gift Idea Pi Dorm Decor Single Print 970 INSTANT DOWNLOAD

- Shop Comfortable Nursing Bras Online – Baby & Me Maternity

- Lularoe TC Tall Curvy Black Solid Leggings Unicorn Stars Cream Blue Red Green 🦄

- 2024 Grand Design Transcend Xplor 245RL

- 20th Century Masters - The Christmas Collection: The Best of 98 Degrees by 98° (Compilation, Christmas Music): Reviews, Ratings, Credits, Song list - Rate Your Music

- Definitely a girl. Fashion look of child girl. Little girl back to school. Informal and non-formal education. Afterschool time. Casual style trends. Kids wardrobe. Hair salon. Beauty for tomorrow Stock Photo
