Developing a Thermodynamical Method for Prediction of Activity

By A Mystery Man Writer
Results of the experimental measurements on the partial molar volume of kerosene used as a medium for dissolving TBP are utilized to determine the activity of TBP in the binary kerosene-TBP solution through the application of Gibbs-Duhem equation. The treatment is based on combination of the experimental data with the thermodynamic values available on the compressibility factor of pure kerosene at room temperature. It is shown that the activity of TBP in kerosene has a positive deviation from ideality with an activity coefficient derived as follows:1) at X TBP ≤ 0.01: γ TBP = 42.530, 2) at the 0.01 X TBP 0.2: 3) at the higher TBP concentrations 0.2 X TBP 0.97: and 4) at TBP Raoultian concentrations 0.97 ≤ X TBP:γ TBP = 1. These quantities can be utilized at temperature closed to 298 K.

PDF) Thermodynamics of extraction of Zn2+ from sulfuric acid media with a mixture of DEHPA and MEHPA
GMD - The Regional Ice Ocean Prediction System v2: a pan-Canadian ocean analysis system using an online tidal harmonic analysis

Separation of Re and Mo from Roasting-Dust Leach-Liquor Using Solvent Extraction Technique by TBP
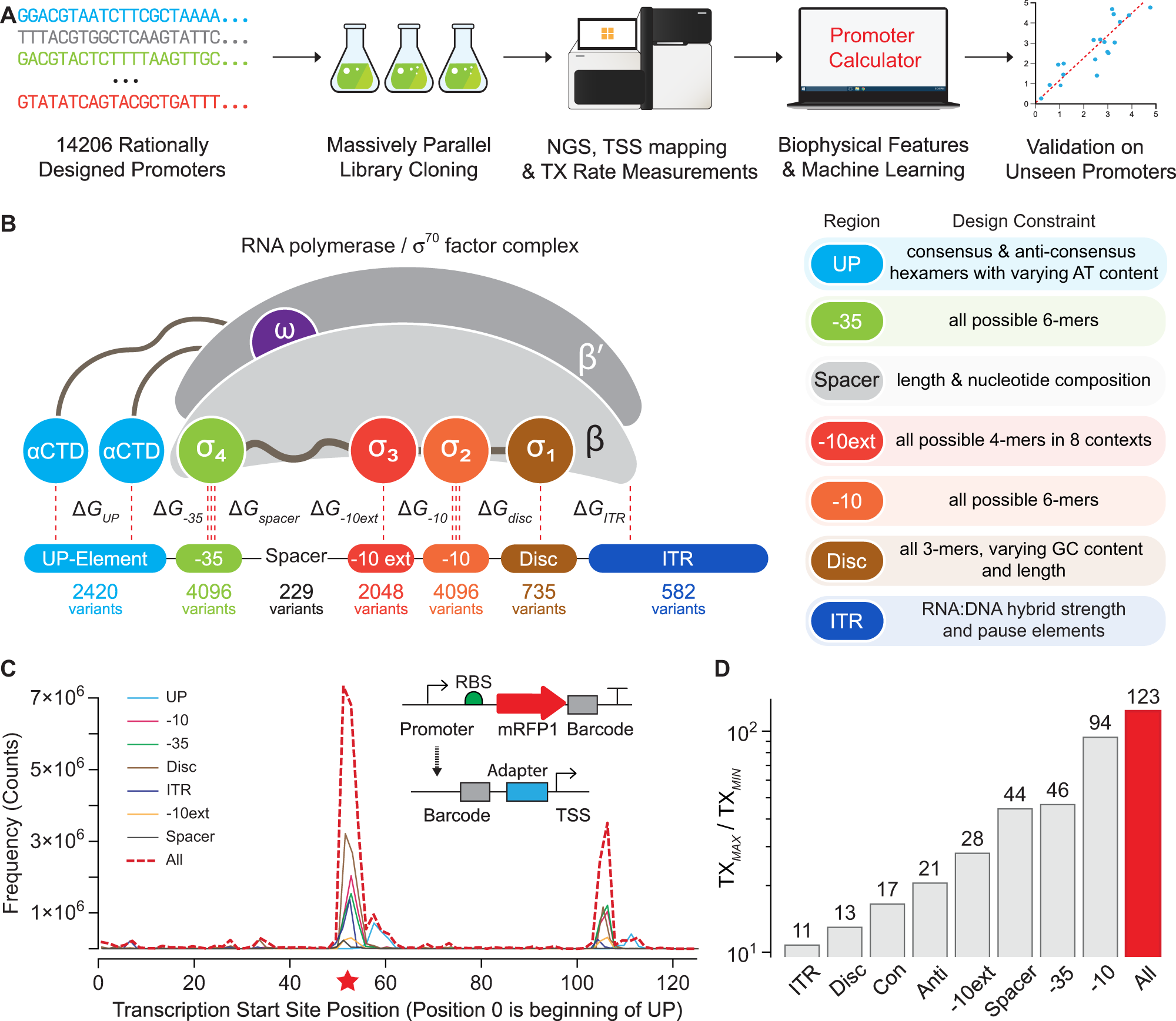
Automated model-predictive design of synthetic promoters to control transcriptional profiles in bacteria
8 Tools to Enable the Future of Chemical Engineering, New Directions for Chemical Engineering

Enthalpy: Energy Transfer in Physical and Chemical Processes - Lesson

E. ALAMDARI, Professor (Associate), PhD, Amirkabir University of Technology, Tehran, TUS, Department of Mining and Metallurgical Engineering

Separation of Re and Mo from Roasting-Dust Leach-Liquor Using Solvent Extraction Technique by TBP
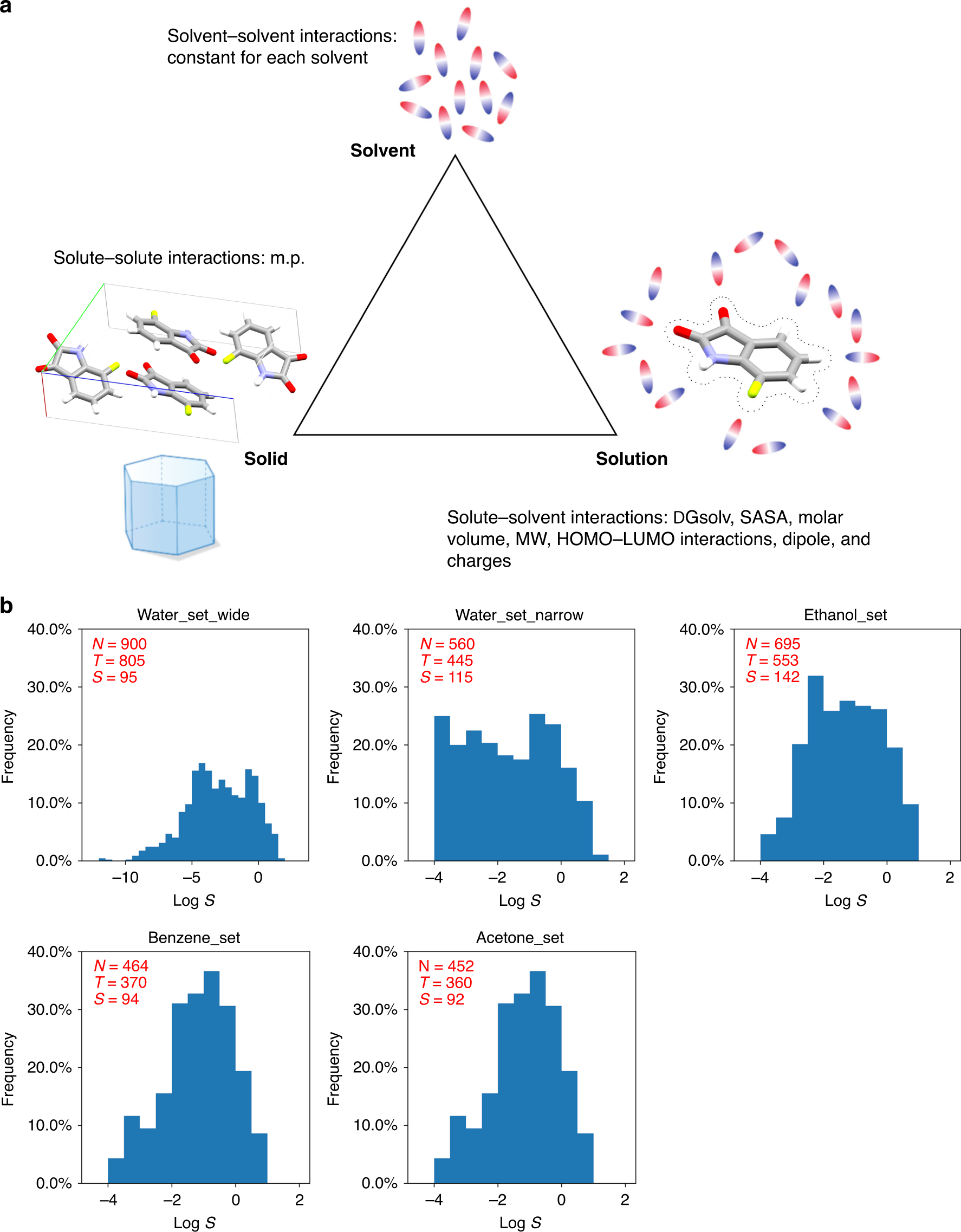
Machine learning with physicochemical relationships: solubility prediction in organic solvents and water
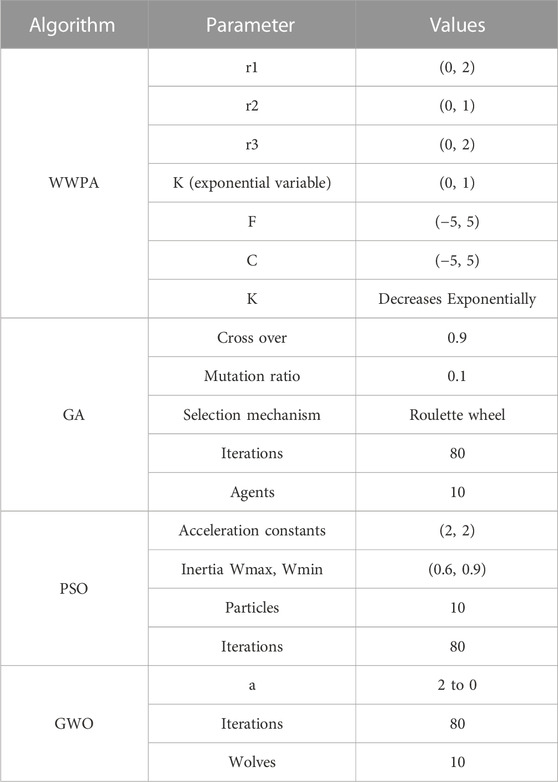
Frontiers Electrical power output prediction of combined cycle power plants using a recurrent neural network optimized by waterwheel plant algorithm

Eskandar Keshavarz Alamdari - PDF Free Download

Separation of Re and Mo from Roasting-Dust Leach-Liquor Using Solvent Extraction Technique by TBP
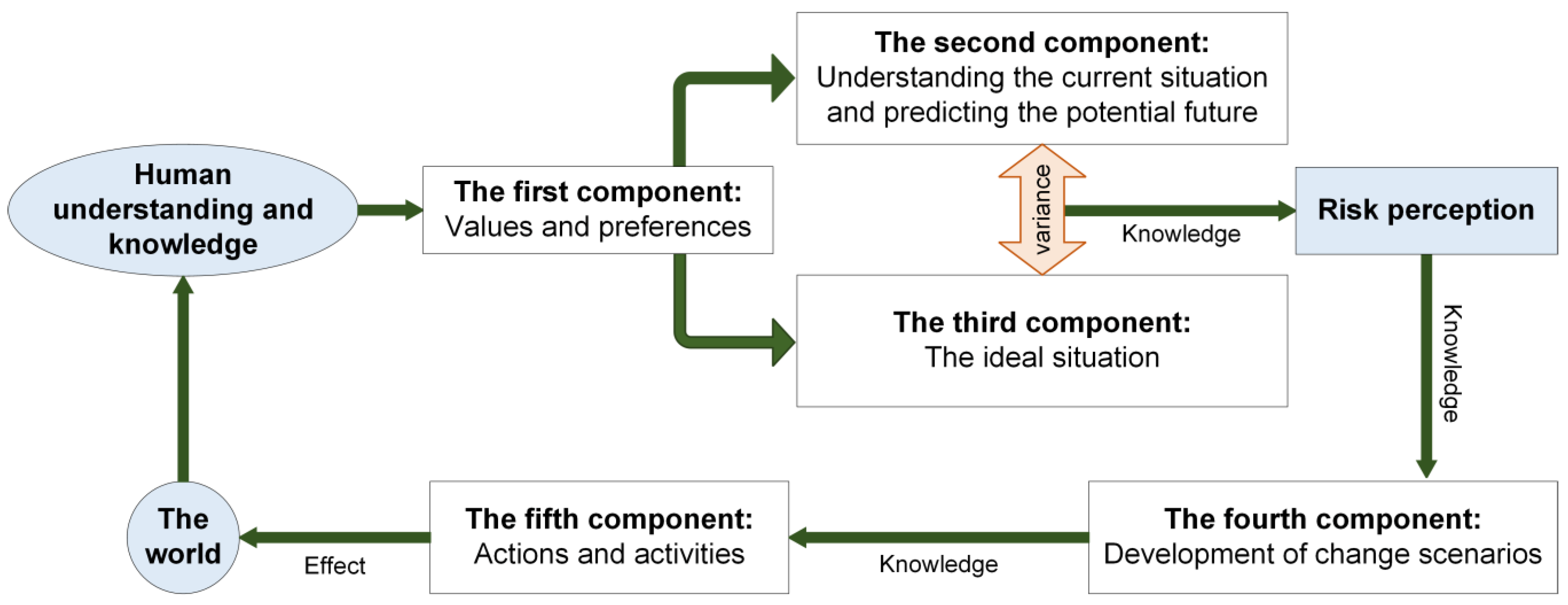
Systems, Free Full-Text
- Compressibility Factor from Redlick-Kwong Equations

- Solved 3) The compressibility factor for a real fluid with

- If Z is a compressibility factor, van der Waals equation at low pressure ..

- Solved] Why is the compressibility factor less than 1 at most conditions?
- Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT





