Useful Forms – UK NEQAS – ICC & ISH
By A Mystery Man Writer

PDF) Assessment of Ki67 in Breast Cancer: Updated Recommendations From the International Ki67 in Breast Cancer Working Group

Global Ring Study to Investigate the Comparability of Total Assay Performance of Commercial Claudin 18 Antibodies for Evaluation in Gastric Cancer - ScienceDirect

Immunohistochemistry should undergo robust validation equivalent to that of molecular diagnostics

Registration - UK NEQAS for H&I

Useful Forms – UK NEQAS – ICC & ISH

UK NEQAS ICC & ISH (@ukneqasicc) / X

PDF) Biomarker testing in oncology – Requirements for organizing external quality assessment programs to improve the performance of laboratory testing: revision of an expert opinion paper on behalf of IQNPath ABSL

Immunohistochemistry Review Of The Technical Test - NordiQC
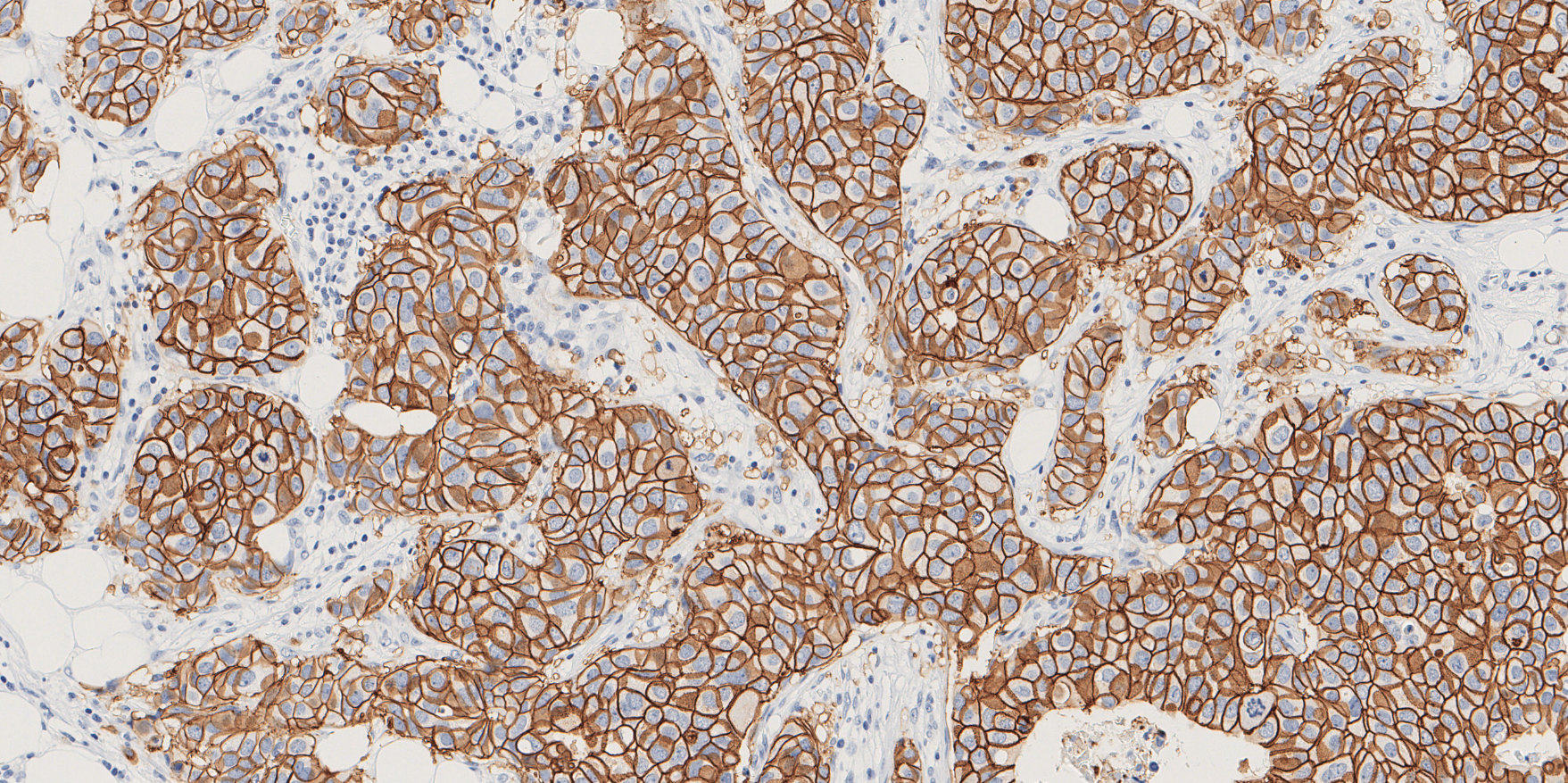
Newsroom Visiopharm

Concordance of HER2-low scoring in breast carcinoma among expert pathologists in the United Kingdom and the republic of Ireland –on behalf of the UK national coordinating committee for breast pathology - The
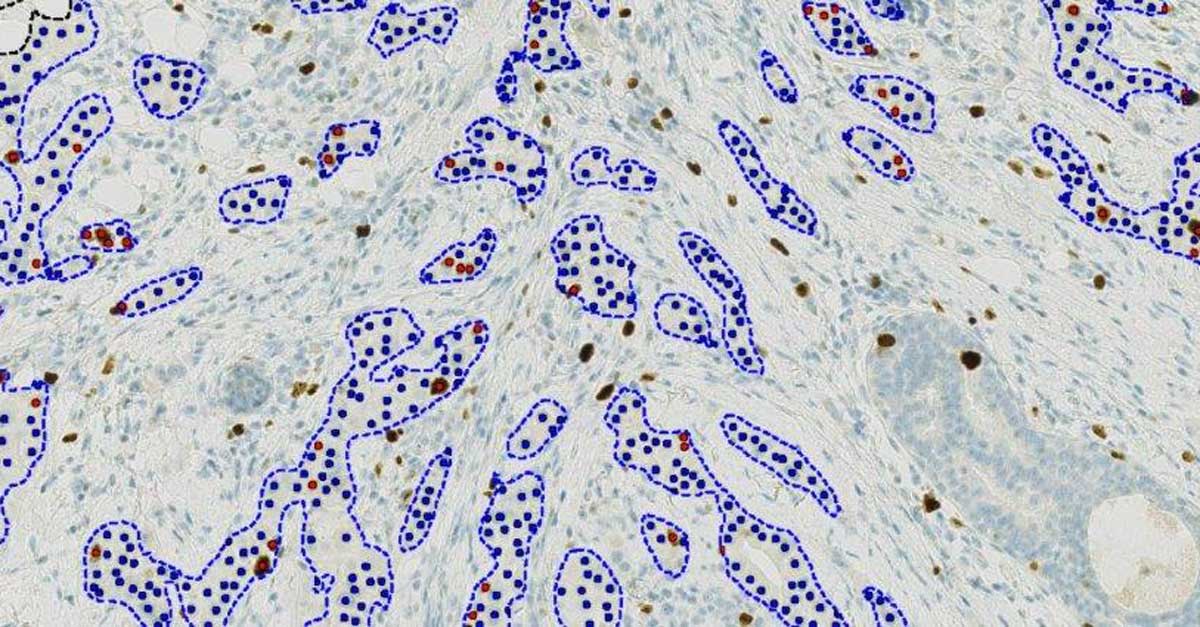
Visiopharm launches IVDR-cleared and fully automated next-generation Ki67 algorithm for IHC biomarker analysis - Visiopharm
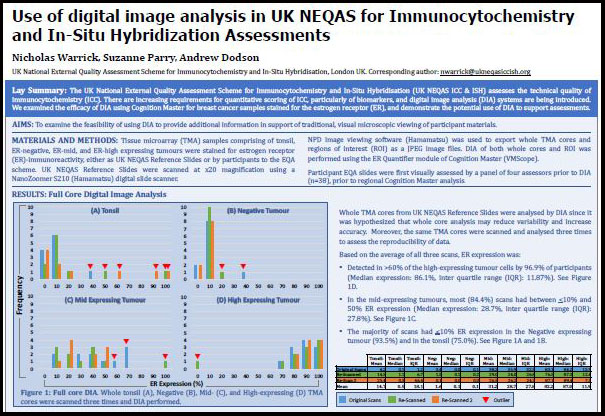
Digital Analysis of UK NEQAS ICC & ISH ER Samples (2020) – UK NEQAS – ICC & ISH

Accreditation and Scope – UK NEQAS – ICC & ISH